Getting Geeky: Malolactic Fermentation
If my last post on malolactic fermentation was a 101-level course, you can consider this an advanced tutorial, complete with biochemistry tie-in.
The conversion of Malic acid to Lactic acid is not technically fermentation.
Fermentation refers to metabolism of a carbon source (like sugar) in the absence of oxygen. When oxygen is not present, the end-product of the metabolic cycle is another organic compound. In the case of the yeast, Saccharomyces, that product is ethanol (alcohol). For certain types of bacteria, including the Lactic Acid Bacteria (LAB) Oenococcus Oeni, that end product could be acetic acid (vinegar). Thus, the presence of LAB in wine is not without risks. The winemaker has to be aware of these risks and manage the wine environment to try to avoid spoilage that would render the wine unmarketable.
So, while technically not a fermentation, people refer to it as such because of the carbon dioxide (CO2) that is released when the bacteria make this conversion. It bubbles.
Malic acid has two carboxyl groups.
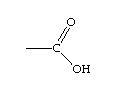 The image is for those of you who remember your organic chemistry. A carboxyl group is acidic, so the more of them you have, the more acidic you are. Lactic acid has the same basic structure as Malic acid, but only one carboxyl group. When the bacteria make the conversion, CO2 is released (bubbles) and the result is a decrease in total acidity.
The image is for those of you who remember your organic chemistry. A carboxyl group is acidic, so the more of them you have, the more acidic you are. Lactic acid has the same basic structure as Malic acid, but only one carboxyl group. When the bacteria make the conversion, CO2 is released (bubbles) and the result is a decrease in total acidity.
Why do the bacteria do this little sleight of hand, when they are not gaining energy from the process?
 While they do not use Malic acid as a fuel source, the effect of DE-carboxylating the organic acid allows them to SAVE overall energy. This has to do with transportation of acid across the cell walls. In order to keep proper intercellular pH, they need to keep exporting acidic protons (I’ve really tried to avoid using this term up ‘til now. The geeks among you will understand). This costs energy. When they do it by means of the acidic conversion described above, they save the energy they otherwise would have expended. So it all makes sense really. Trust me.
While they do not use Malic acid as a fuel source, the effect of DE-carboxylating the organic acid allows them to SAVE overall energy. This has to do with transportation of acid across the cell walls. In order to keep proper intercellular pH, they need to keep exporting acidic protons (I’ve really tried to avoid using this term up ‘til now. The geeks among you will understand). This costs energy. When they do it by means of the acidic conversion described above, they save the energy they otherwise would have expended. So it all makes sense really. Trust me.
Also, I’d like to explain a little more about the buttery aroma I mentioned in my primer on malolactic fermentation. In fact, this is an accidental byproduct. In the presence of a small amount of oxygen, a chemical compound is formed called diacetyl. The precursor to diacetyl kind of leaks out of the citric acid metabolic pathway. This chemical compound really smells like butter. In high concentrations, it reminds me of movie popcorn buttery liquid. I definitely don’t want it in the red wines we produce. Luckily, when you leave well enough alone, the bacteria (and sometimes some remaining yeast cells too), will grab onto the diacetyl and convert it to organic compounds that have no sensory impact on the wine. Once you add sulfites to the wine, these microorganisms die off, and any remaining diacetyl will be preserved. I typically wait 10-14 days after ML finishes before adding sulfites to ensure that this process is complete.
So, all these things are going on right now in our new wines. It is why we are so diligent about tasting and analysis. And it is why we don’t feel much like we can take a break and relax until everything is settled.